COVID-19 Disease Research

COVID-19 disease immune monitoring and spatial tissue profiling research
Standard BioTools™ CyTOF™ technology with the Maxpar™ Direct Immune Profiling Assay provides best-in-class immune monitoring with the cost, flexibility and consistency needed for standardized COVID-19 disease immune monitoring research. Imaging Mass Cytometry™ technology adds the capability of spatial visualization of immune response in tissue samples. This newly developed technology enables the study of clinical outcomes and changes in inflammatory or immune function directly from whole blood samples or tissues.
Many questions remain unanswered in COVID-19 disease research:
- What immunologic features and molecular mechanisms are involved in COVID-19 severity?
- Can those at risk of severe symptoms be identified early?
- What interventions can help to reduce severity, and why do they work?
- What can immune infiltrates tell us about the aftermath of infection?
- Which vaccine candidates are most effective?
Standard BioTools mass cytometry and Imaging Mass Cytometry systems, powered by CyTOF technology, are being used by researchers around the world to help find answers to these and many other important questions to understand the unprecedented COVID-19 disease.
Learn how we can support COVID-19 disease research in many different ways.
 |
|
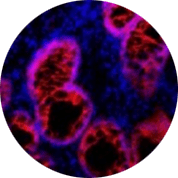 |
High-plex spatial visualization and phenotyping of early COVID-19 studies |
 |
Immune profiling to better understand the immune response to COVID-19
Obtaining an information-rich profile of the features and functionality of a patient’s immune system during the course of infection or in response to therapy is of critical importance.
The Maxpar Direct Immune Profiling System provides an ideal solution for such longitudinal immune monitoring studies with either whole blood or PBMC samples.

The convenient single-tube format and easy workflow allow many patient samples to be processed in a single day.
Processed samples can be frozen and saved or shipped for collection on a Helios™ mass cytometer at a later time or distant location.
The pushbutton analytic software, Maxpar Pathsetter, identifies and enumerates 37 immune cell populations in 5 minutes or less and requires no special expertise to use.
View the high-quality data delivered by the system.
Resources
Publications overviews
Spotlight articles
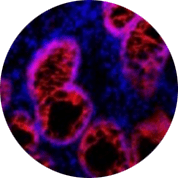
High-plex spatial visualization and phenotyping of early COVID-19 studies
High-plex spatial profiling of the host immune response can enable deep biological insight using the Hyperion™ Imaging System to perform Imaging Mass Cytometry (IMC™). Visualization and quantification of immune infiltrates in COVID-19 tissue samples are becoming an important IMC application.
Researchers using IHC can benefit from the high-dimensional spatial visualization that IMC offers
An early COVID-19 publication showing the histopathology of lung tissue from a critically ill patient was submitted by Luo et al. In Clinical pathology of critical patient with novel coronavirus pneumonia (COVID-19) (2020), the authors describe their research to understand the pathogenesis and severity of the disease.
This team utilized immunohistochemistry (IHC) to positively identify several markers in immune infiltrates in a lung biopsy sample. The results suggested the general spatial context of the cells present focally in lung interstitium and near blood vessels.
A short summary of the publication results is included in the Standard BioTools article Imaging Mass Cytometry Publication Review. After the summary, commentary from Standard BioTools introduces how researchers using IHC could benefit from using the Hyperion Imaging System to characterize lung tissue from COVID-19 or other diseases. Certainly, IHC results demonstrate how this approach can be used to spatially assess immune infiltration. However, by leveraging IMC technology, deeper insights from profiling many markers in a single scan could be achieved while minimizing the use of precious disease tissue samples. The article includes an example of lung tissue visualized using IMC platforms to look at multiple markers in the same scan.
Highlights
IHC validation of IMC results
Two recent IMC publications include information validating IMC work to IHC. In The single-cell pathology landscape of breast cancer (Jackson et al., Nature, 2020), extended data Figure 2 compares IMC and IHC technologies and demonstrates reproducibility of quantified stains in sections of the same tumor core. Similarly, as part of the development of their 40-marker IMC panel, Ijsselsteijn et al. describe how they used IHC to test antibody specificity before and after metal conjugation as well as to determine the optimal antigen retrieval conditions (A 40-marker panel for high dimensional characterization of cancer immune microenvironments by Imaging Mass Cytometry, Frontiers in Immunology, 2019). Both publications observe comparable immunodetection patterns between IHC and IMC technology. This indicates that IHC can be used as a convenient tool for the low-cost validation of Imaging Mass Cytometry antibodies.
See IMC publication bibliography
The first published study demonstrating the use of IMC for COVID-19 disease research
In Inflammatory response cells during acute respiratory distress syndrome in patients with coronavirus disease 2019 (COVID-19) (Annals of Internal Medicine, 2020), Zhang et al. describe their use of a high-dimensional Imaging Mass Cytometry panel that allowed for an unbiased exploratory approach to immune infiltration in two COVID-19 patients: a 94-year-old who died of cardiac arrest and a 65-year-old who died of pneumonia complications. In this article you can see a representative example of images showing selected markers from their panel, all scanned simultaneously from a single lung section.
Generation of a single-cell atlas for terminal bronchioles
A study that highlights the importance of early diagnosis might also be applied to COVID-19 research. In mild to moderate chronic obstructive pulmonary disease (COPD), the smallest conducting airways in the lungs, terminal bronchioles, become severely reduced before emphysematous destruction of the lung alveolar surface area. Booth et al. performed a cross-sectional analysis using IMC technology to generate a single cell atlas for these pulmonary regions. By applying machine learning, the team observed an activated immune response with increased interaction between innate and adaptive immune cells within the terminal bronchioles of mild and moderate COPD patients (An in-situ single cell atlas of the terminal bronchioles in chronic obstructive pulmonary disease by Imaging Mass Cytometry, American Journal of Respiratory and Critical Care Medicine, 2020).
Other valuable resources for understanding how IMC platforms can be used in COVID-19 disease research are located below.
Other Imaging Mass Cytometry references to support COVID-19 research: The information and links here describe how IMC technology might be incorporated into infectious disease studies, such as those for COVID-19.
In this publication a team from Yale discusses how it used IMC technology to investigate Zika virus infection. Uraki, R. et al. “Aedes aegypti AgBR1 antibodies modulate early Zika virus infection of mice.” Nature Microbiology 4 (2019): 948–955.
Watch this 10-minute presentation by one of the authors of the Nature Microbiology publication, Ruth Montgomery, PhD, Professor of Internal Medicine, Director of the Yale CyTOF Facility and Associate Dean for Scientific Affairs. In this webinar, “Profiling of COVID-19 Patient Immune Responses,” Montgomery discusses her COVID-19 research.
Watch this webinar by Tom Ashhurst, PhD, High‐Dimensional Cytometry Specialist at the Sydney Cytometry Facility. In the webinar titled “Mapping Dynamic Immunity Across Time, Space, and Disease State,” Ashhurst describes how he uses mass cytometry and Imaging Mass Cytometry to better understand viral diseases of the central nervous system.
Get more information
For Research Use Only. Not for use in diagnostic procedures. Patent and License Information: www.standardbio.com/legal/notices. Trademarks: www.standardbio.com/legal/trademarks. Any other trademarks are the sole property of their respective owners. ©2025 Standard BioTools Inc. All rights reserved.
