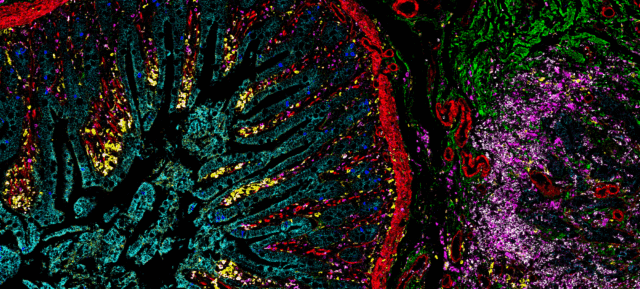
To kick off the first session of our current IMC Forum series on data analysis, a Senior Field Applications Scientist and Imaging Mass Cytometry expert walks you through the benefits you’ll see from using IMC technology.
Now that you’ve decided that multiplexed imaging can contribute essential spatial information to your biological research, you’re ready to create beautiful images using Imaging Mass Cytometry™ (IMC™) technology. Before you start, take a quick look at some recommendations from Wendell Smith, Standard BioTools™ Senior Field Applications Scientist and IMC expert. Wendell has a background in immunology and received his PhD from the University of California San Diego working in the laboratory of Flossie Wong-Staal, PhD. He went on to complete his postdoctoral studies at the La Jolla Institute under Carl Ware, PhD, specializing in virology and immunology.
To kick off Stepping Through the Pipeline: Recommendations and Considerations for In-Depth Analysis, the first session of our current IMC Forum series, Wendell walks you through the benefits you’ll see from using IMC technology, considerations for ensuring the cleanest images and what you can do with cell segmentation to have the utmost confidence in your results.
Why IMC?
IMC platforms enable detailed assessment of the tissue microenvironment using 40-plus markers simultaneously at subcellular resolution on a single slide. Its use is well established in publications and in over 15 clinical trials and continues to grow as a trusted method for high-plex imaging of the tissue and tumor microenvironment.
IMC isn’t a fluorophore-based technology; like other platforms, it uses antibodies to identify proteins, but those antibodies are conjugated to metals, which produce distinct signals for precise detection of individual proteins. Fluorophores have overlap in the emission spectrum, which means that the more fluorophores you add, the more complex your process becomes. Spectral overlap also means the use of fluorophores makes it difficult to adjust your panels. IMC technology doesn’t have emission of light to consider, so adjusting panels is a much more streamlined process.
One way to get around the issue of spectral overlap and avoid compensation is to minimize the number of fluorophores used. However, for high-plex imaging (or for imaging more than four or five markers), numerous fluorophores mean numerous cycling scans, making analysis more time-consuming and resulting in multiple chemical exposures to your tissue. The IMC workflow is simple and familiar. Standard BioTools recommends optimizing your panel and titration of antibodies, then a simple staining of all the antibodies on your slide at once followed by acquisition and data analysis.
The inherent background fluorescence in tissues from red blood cells, collagen and flavins along with other native components can significantly affect the signal-to-noise ratio. This can mask important signals in tissues and present challenges when trying to distinguish cell populations or complex architecture.
A key advantage of the IMC workflow is the lack of autofluorescence. IMC technology uses rare-earth metals instead of fluorophores, meaning the natural fluorescence that occurs in biological structures is avoided and other background is very limited since heavy metals do not naturally exist in biological tissues. This gives you much cleaner, reproducible data.
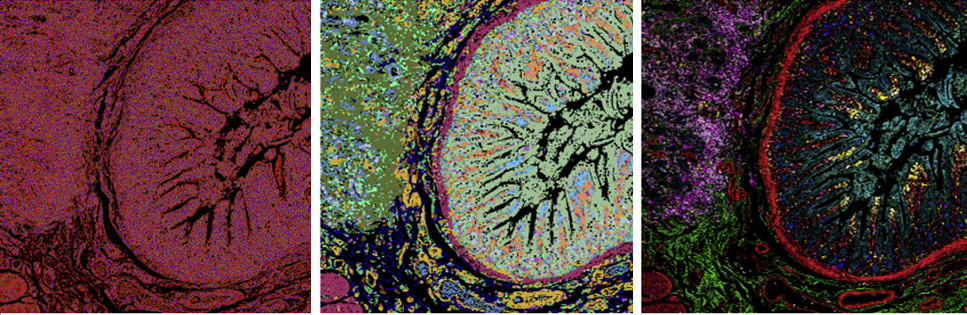
Pre-analysis considerations
All imaging analysis contains artifacts and noise to varying degrees. Here are some causes of noise, and some of our recommended solutions:
- Suboptimal antibody titration: Nonspecific binding of antibodies is a natural phenomenon and can happen with any antibody-based technology. Optimizing the amount of antibody to ensure you have the best signal-to-noise ratio results in a good dynamic range of around 1 x 105.
- Channel crosstalk: Standard BioTools metals are selected because they’re very pure, with about a 98% purity cutoff. However, if you have too high of an antibody concentration crosstalk can still occur. Titration also ensures crosstalk is minimized.
- Oxidization: It’s crucial to take good care of your antibodies, ensuring that they don’t oxidize. When you use antibodies, make sure to cap them and return them to the fridge when you’re done.
Another critical step in analysis is cell segmentation, in which clear cell boundaries are identified. Cell segmentation is essential for quantitative, single-cell analysis in which cell types, cellular functions and intra- and intercellular processes can be defined. Spending time ensuring your segmentation is correct will result in trustworthy analysis. Our Maxpar™ IMC Cell Segmentation Kit contains three individual plasma membrane markers conjugated to 195Pt, 196Pt and 198Pt. These markers can be added to existing panels for improved nucleus and plasma membrane demarcation, enabling cell segmentation across multiple tissue types without taking channels away from your current panel. You can also refer to our IMC Forum on Current Methods and Future Views for Effective Cell Segmentation in High-Plex Imaging to learn more.
Analysis
There are a number of options for higher-order data analysis, including dimensionality reduction and clustering, neighborhood analysis, phenotype heat map and manual gating. Some of the tools you can use include histoCAT, ImaCytE, FlowJo and Visiopharm® Phenoplex™ analysis software. For those who have programming skills, data preprocessing with R is a good alternative to visualize your data. Learn more about commonly used open source and commercial software algorithms and analytical tools and how our FAS team can help you decide on an approach to meet your research goals by watching the second session in our IMC Forum on data analysis.
Summary
- Consider noise and how it can affect your data
- Careful segmentation is key to achieving high-quality analysis
- Make sure you find the right algorithm for the biological question(s) at hand
- Plan data mining and analysis strategies to get the most out of the vast amount of information our platform enables