New panels that focus on cell signaling and metabolism help researchers interrogate the tumor microenvironment like never before
The tumor microenvironment (TME) is the cellular and molecular ecosystem that surrounds a tumor. It’s composed of many moving parts, such as cancer cells, stromal tissue and the extracellular matrix, a network of proteins and carbohydrates that provides structural support and controls the development and function of tissues. The TME is constantly changing, depending on its location, the tumor stage and even patient characteristics, so monitoring and interpreting its variation remains a complex endeavor.
Where researchers once believed cancer was a cell and gene expression disorder, they now consider it to be a TME disease, meaning that gaining a better understanding of the tumor microenvironment is key to advancing scientific understanding of cancer biology. The TME plays a key role in the progression and metastasis of cancer: Interactions between cancer cells and components of the tumor microenvironment support invasion into healthy tissues, which correlates with poor treatment response and prognosis. In addition, cancer growth and metastasis are affected by immune cell and cancer cell interactions in the TME.
Shedding light on the interactions between cancer cells and the TME helps researchers better understand the progression of cancer and explore biological factors that may be associated with therapeutic research questions. Standard BioTools™ modular Imaging Mass Cytometry™ (IMC™) antibody panels simplify spatial profiling of the TME, detecting key proteins that identify cell metabolic states and changes in signaling pathways.
Cell signaling dynamics
Signaling pathways are networks of molecules that relay signals inside of a cell, allowing that cell to respond to changes in its environment. Signaling pathways in tumor cells may be significantly different from the pathways of healthy cells, supporting uncontrolled proliferation and survival instead of executing and controlling important cellular survival and growth processes.
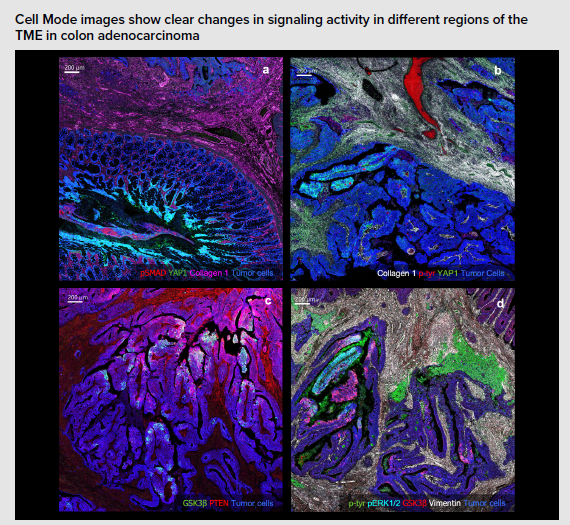
Understanding the activation states of cell signaling pathways in the TME can provide more information on the progression of cancer and also support research aimed at understanding immunotherapy development. The Human Cell Signaling IMC Panel, 7 Antibodies contains essential markers for the identification and study of major signaling pathways in human cells, including seven pathologist-verified antibodies – EGFR, pSMAD, GSK3β, p-tyrosine, YAP1, pERK1/2 and PTEN – that enable the assessment of the EGFR, TGF-β, Wnt and Hippo signaling, and MAPK and PI3K/Akt pathways.
These pathways are fundamental regulators of key cellular processes:
- Epidermal growth factor receptor (EGFR) signaling plays a key role in tumorigenesis across multiple cancer types. Aberrant EGFR expression drives cancer progression, particularly in non-small-cell lung cancer, glioblastoma, colorectal cancer, gastric cancer, and head and neck cancers.
- Transforming growth factor-β (TGF-β) signaling is essential in processes such as chemotaxis, apoptosis, differentiation, growth and cell migration. It can also drive cancer progression through epithelial–mesenchymal transition, the modulation of factor expression and evasion of immune responses.
- WNT signaling regulates cell proliferation, differentiation and stemness, and plays a critical role in tumorigenesis, cancer progression and therapeutic resistance
- Hippo signaling controls organ size and tissue homeostasis; its aberrant expression contributes to cancer development
- The mitogen-activated protein kinase (MAPK) pathway, considered a critical regulator of intracellular and extracellular signaling, plays a significant role in the emergence of chemoresistance
- The PI3K/AKT/mTOR pathway assists with the regulation of events such as cell proliferation, cell growth and survival, and metabolism. Dysregulation can lead to tumor growth, poorer survival and resistance to treatment
The combination of the Hyperion™ XTi Imaging System and targeted IMC panels helps visualize the heterogeneity and spatial distribution of signaling pathways in tumor cell populations. Seen below, used on colon adenocarcinoma:

Metabolic reprogramming
Cell metabolism is the series of biochemical reactions that enable cells to produce the energy required for their survival and maintenance. Cancer cells are particularly metabolically adaptable, undergoing metabolic reprogramming to sustain rapid proliferation, survive in harsh microenvironments and resist therapies. This complexity means that assessing the metabolic states of cancer cells is a difficult undertaking – one that requires flexible, high-throughput tools.
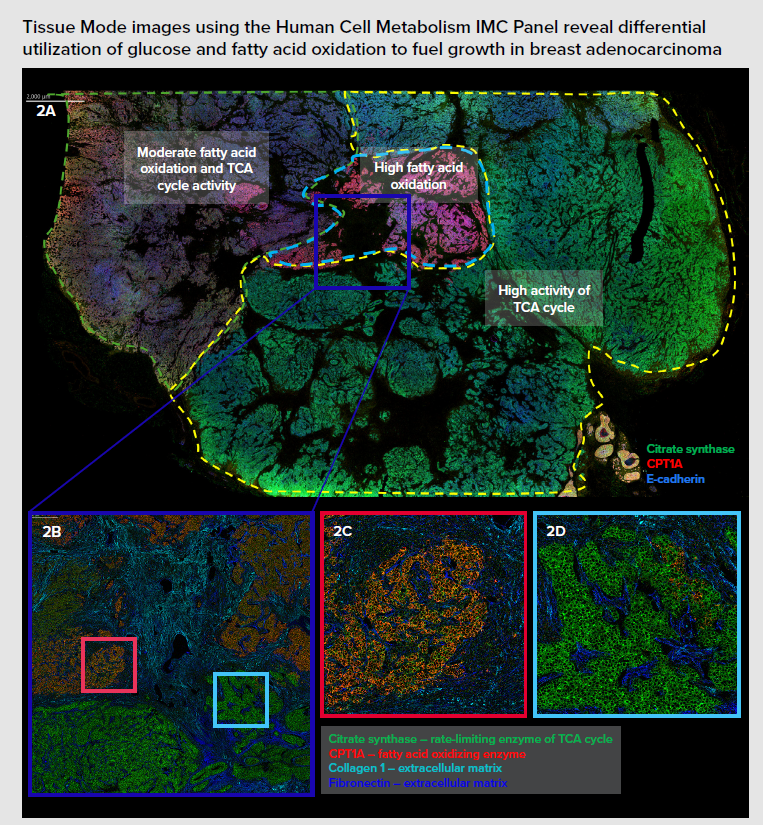
Monitoring cell metabolism and revealing the positioning of tumor and immune cells can shed light on alterations in metabolic activity and cellular energy production, which is essential for research into elucidating disease progression advancing the development of immunotherapies. The Human Cell Metabolism IMC Panel, 7 Antibodies, provides markers for the identification and study of six metabolic pathways: glucose metabolism, fatty acid oxidation, lactate metabolism, the tricarboxylic acid (TCA) cycle, oxidative phosphorylation and mTOR signaling. The panel includes seven pathologist-verified antibodies – GLUT1, citrate synthase, ATP5A, CPT1A, LDHA, p4E-BP1 and pS6 – that can be stained and acquired simultaneously.
A metabolic pathway is a series of connected chemical reactions that feed one another:
- Glucose metabolism is a biochemical process that involves the absorption, transformation, synthesis, degradation and use of glycans, providing fuel for cellular activities
- Fatty acid oxidation is the metabolic process occurring primarily in mitochondria in which fatty acids are broken down to produce adenosine triphosphate (ATP)
- The metabolism of lactate, a molecule primarily known as a byproduct of glycolysis, plays a role in energy supply, glycemia maintenance, cerebral metabolism and signaling
- The TCA cycle is a series of biochemical reactions that release the energy stored in nutrients through acetyl-CoA oxidation; the energy released is available in the form of ATP
- Oxidative phosphorylation is the metabolic pathway in which cells use enzymes to oxidize nutrients, releasing chemical energy and producing ATP
- The mammalian target of rapamycin (mTOR) signaling pathway serves as a key regulator of cell metabolism, growth, proliferation and survival
Investigating processes that regulate energy production can support research into metabolic dysfunction linked to cardiovascular diseases, cancer, neurological conditions and age-related diseases. Using the Hyperion XTi Imaging System and IMC panels can reveal significant insights into the spatial organization and metabolic profile of cells across cancer tissue, as seen in the example below on breast adenocarcinoma:

The emergence of new technologies has allowed researchers to spatially resolve and interrogate the tumor microenvironment like never before. Modular IMC panels enable comprehensive spatial profiling, revealing the interconnected roles signaling pathways play in promoting tumor survival and resistance to therapies. Their application uncovers cell signaling dynamics in the TME, potentially empowering researchers with insights that can inform and inspire future advances in cancer research.